A chemical bond is a lasting attraction between these atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds. Therefore, the electromagnetic force plays a major role in determining the internal properties of most objects encountered in daily life.
Intramolecular bonds
- Ionic bond. An ionic bond is a chemical bond, in which one or more electrons are wholly transferred from an atom of one element to the atom of the other, and the elements are held together by the force of attraction due to the opposite polarity of the charge. This type of chemical bond is typical between elements with a large electronegativity difference.
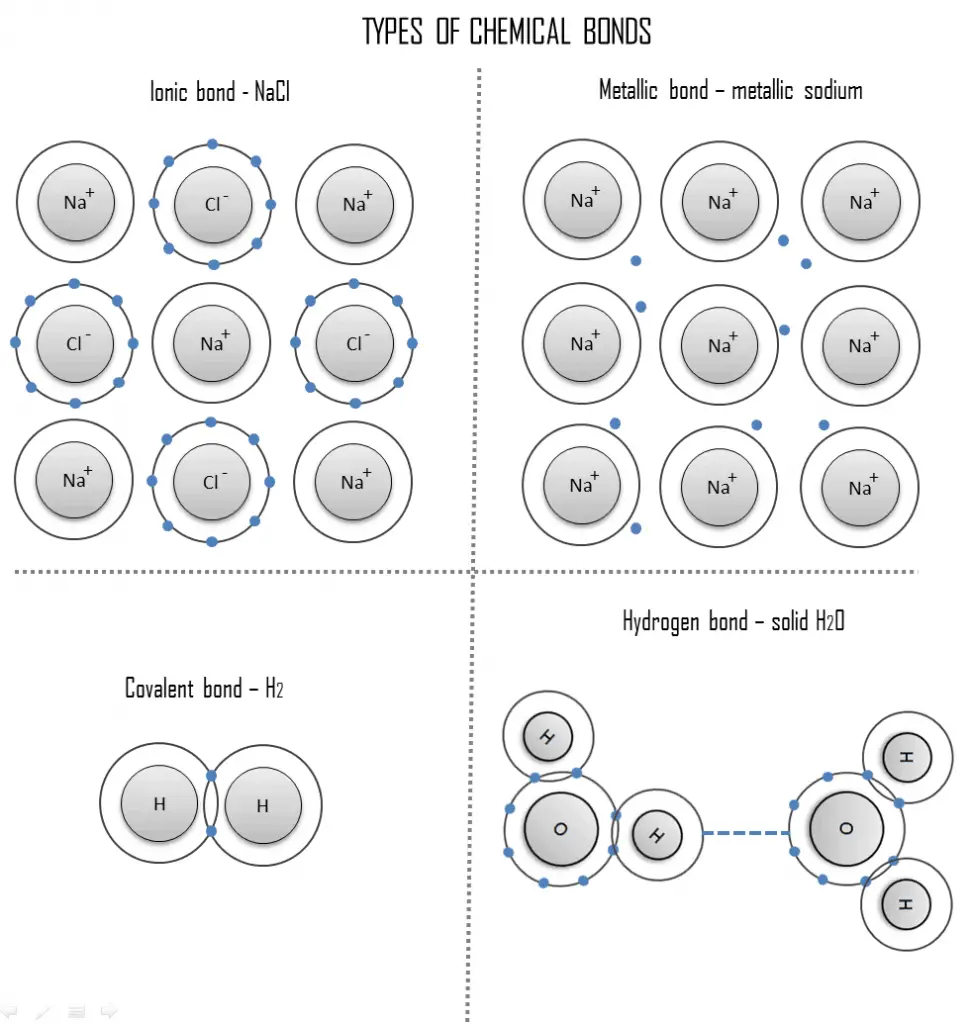
- Covalent bond. A covalent bond is a chemical bond formed by shared electrons. Valence electrons are shared when an atom needs electrons to complete its outer shell and can share those electrons with its neighbor. The electrons are then part of both atoms and both shells are filled.
- Metallic bond. A metallic bond is a chemical bond, in which the atoms do not share or exchange electrons to bond together. Instead, many electrons (roughly one for each atom) are more or less free to move throughout the metal, so that each electron can interact with many of the fixed atoms.
Covalent Bond
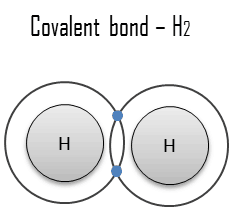 A covalent bond is a chemical bond formed by shared electrons. Valence electrons are shared when an atom needs electrons to complete its outer shell and can share those electrons with its neighbor. The electrons are then part of both atoms and both shells are filled. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
A covalent bond is a chemical bond formed by shared electrons. Valence electrons are shared when an atom needs electrons to complete its outer shell and can share those electrons with its neighbor. The electrons are then part of both atoms and both shells are filled. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
The simplest and most common type is a single bond in which two atoms share two electrons. Other types include the double bond (e.g. H2C=CH2), the triple bond, one- and three-electron bonds, the three-center two-electron bond and three-center four-electron bond.
Covalency is greatest between atoms of similar electronegativities. Thus, covalent bonding does not necessarily require that the two atoms be of the same elements, only that they be of comparable electronegativity (i.e. elements that lie near one another in the periodic table). There is no precise value that distinguishes ionic from covalent bonding, but an electronegativity difference of over 1.7 is likely to be ionic while a difference of less than 1.7 is likely to be covalent.
Metallic Bond
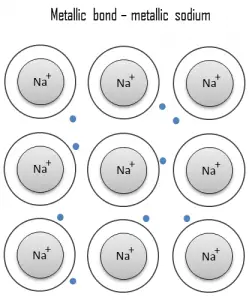 A metallic bond is a chemical bond, in which the atoms do not share or exchange electrons to bond together. Instead, many electrons (roughly one for each atom) are more or less free to move throughout the metal, so that each electron can interact with many of the fixed atoms. The free electrons shield the positively charged ion cores from the mutually repulsive electrostatic forces that they would otherwise exert upon one another; consequently, the metallic bond is nondirectional in character. Metallic bonding is found in metals and their alloys. The free movement or delocalization of bonding electrons leads to classical metallic properties such as luster (surface light reflectivity), electrical and thermal conductivity, ductility, and high tensile strength.
A metallic bond is a chemical bond, in which the atoms do not share or exchange electrons to bond together. Instead, many electrons (roughly one for each atom) are more or less free to move throughout the metal, so that each electron can interact with many of the fixed atoms. The free electrons shield the positively charged ion cores from the mutually repulsive electrostatic forces that they would otherwise exert upon one another; consequently, the metallic bond is nondirectional in character. Metallic bonding is found in metals and their alloys. The free movement or delocalization of bonding electrons leads to classical metallic properties such as luster (surface light reflectivity), electrical and thermal conductivity, ductility, and high tensile strength.
Metal is a material (usually solid) comprising one or more metallic elements (e.g., iron, aluminum, copper, chromium, titanium, gold, nickel), and often also nonmetallic elements (e.g., carbon, nitrogen, oxygen) in relatively small amounts. The unique feature of metals as far as their structure is concerned is the presence of charge carriers, specifically electrons. This feature is given by the nature of metallic bond. The electrical and thermal conductivities of metals originate from the fact that their outer electrons are delocalized.
We hope, this article, Covalent Bond vs Metallic Bond, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about materials and their properties.