This article contains comparison of key thermal and atomic properties of krypton and xenon, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Krypton vs Xenon.
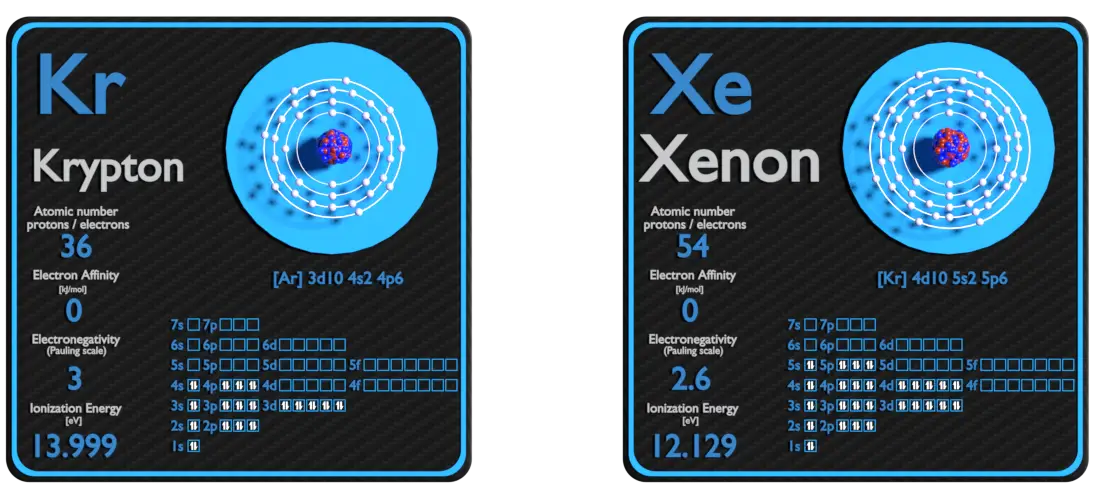
Krypton and Xenon – About Elements


Source: www.luciteria.com
Krypton and Xenon – Applications
Krypton
Krypton’s multiple emission lines make ionized krypton gas discharges appear whitish, which in turn makes krypton-based bulbs useful in photography as a white light source. Krypton is used in some photographic flashes for high speed photography. Krypton is also used in MRI/CT techniques.
Xenon
Xenon is useful in the following applications. The white flash of light produced by xenon makes it suitable for usage in strobe lights and to power ruby lasers. Xenon is used in light-emitting devices called xenon flash lamps, used in photographic flashes and stroboscopic lamps.
Krypton and Xenon – Comparison in Table
| Element | Krypton | Xenon |
| Density | 0.00375 g/cm3 | 0.0059 g/cm3 |
| Ultimate Tensile Strength | N/A | N/A |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | N/A | N/A |
| Mohs Scale | N/A | N/A |
| Brinell Hardness | N/A | N/A |
| Vickers Hardness | N/A | N/A |
| Melting Point | -248 °C | -157.36 °C |
| Boiling Point | -248.7 °C | -153.22 °C |
| Thermal Conductivity | 0.00949 W/mK | 0.00565 W/mK |
| Thermal Expansion Coefficient | N/A | N/A |
| Specific Heat | 0.248 J/g K | 0.158 J/g K |
| Heat of Fusion | 1.638 kJ/mol | 2.297 kJ/mol |
| Heat of Vaporization | 9.029 kJ/mol | 12.636 kJ/mol |




