Magnesium alloys are mixtures of magnesium and other alloying metal, usually aluminium, zinc, silicon, manganese, copper and zirconium. Since the most outstanding characteristic of magnesium is its density, 1.7 g/cm3, its alloys are used where light weight is an important consideration (e.g., in aircraft components). Magnesium has the lowest melting point (923 K (1,202 °F)) of all the alkaline earth metals. Pure magnesium has an HCP crystal structure, is relatively soft, and has a low elastic modulus: 45 GPa. Magnesium alloys have also a hexagonal lattice structure, which affects the fundamental properties of these alloys. At room temperature, magnesium and its alloys are difficult to perform cold working due to the fact plastic deformation of the hexagonal lattice is more complicated than in cubic latticed metals like aluminium, copper and steel. Therefore, magnesium alloys are typically used as cast alloys. Despite the reactive nature of the pure magnesium powder, magnesium metal and its alloys have good resistance to corrosion.
Magnesium alloys are mixtures of magnesium and other alloying metal, usually aluminium, zinc, silicon, manganese, copper and zirconium. Since the most outstanding characteristic of magnesium is its density, 1.7 g/cm3, its alloys are used where light weight is an important consideration (e.g., in aircraft components). Magnesium has the lowest melting point (923 K (1,202 °F)) of all the alkaline earth metals. Pure magnesium has an HCP crystal structure, is relatively soft, and has a low elastic modulus: 45 GPa. Magnesium alloys have also a hexagonal lattice structure, which affects the fundamental properties of these alloys. At room temperature, magnesium and its alloys are difficult to perform cold working due to the fact plastic deformation of the hexagonal lattice is more complicated than in cubic latticed metals like aluminium, copper and steel. Therefore, magnesium alloys are typically used as cast alloys. Despite the reactive nature of the pure magnesium powder, magnesium metal and its alloys have good resistance to corrosion.
Aluminium is the most common alloying element. Aluminium, zinc, zirconium, and thorium promote precipitation hardening: manganese improves corrosion resistance; and tin improves castability.
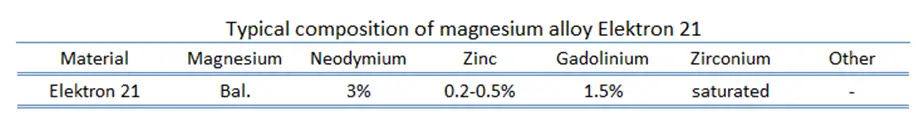
Elektron 21 – UNS M12310
In general, Elektron is the registered trademark of a wide range of magnesium alloys manufactured by a British company Magnesium Elektron Limited. Elektron 21, designated by UNS M12310, is one of alloys with excellent corrosion resistance and castability. Cast products possess a fine-grained microstructure and pressure tightness. This alloy can be easily machined. Application include motorsports and aerospace, since it possess high strength, light weight and it has excellent vibration damping characteristics.
We hope, this article, Composition of Magnesium Alloys, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about materials and their properties.