This article contains comparison of key thermal and atomic properties of arsenic and antimony, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Arsenic vs Antimony.
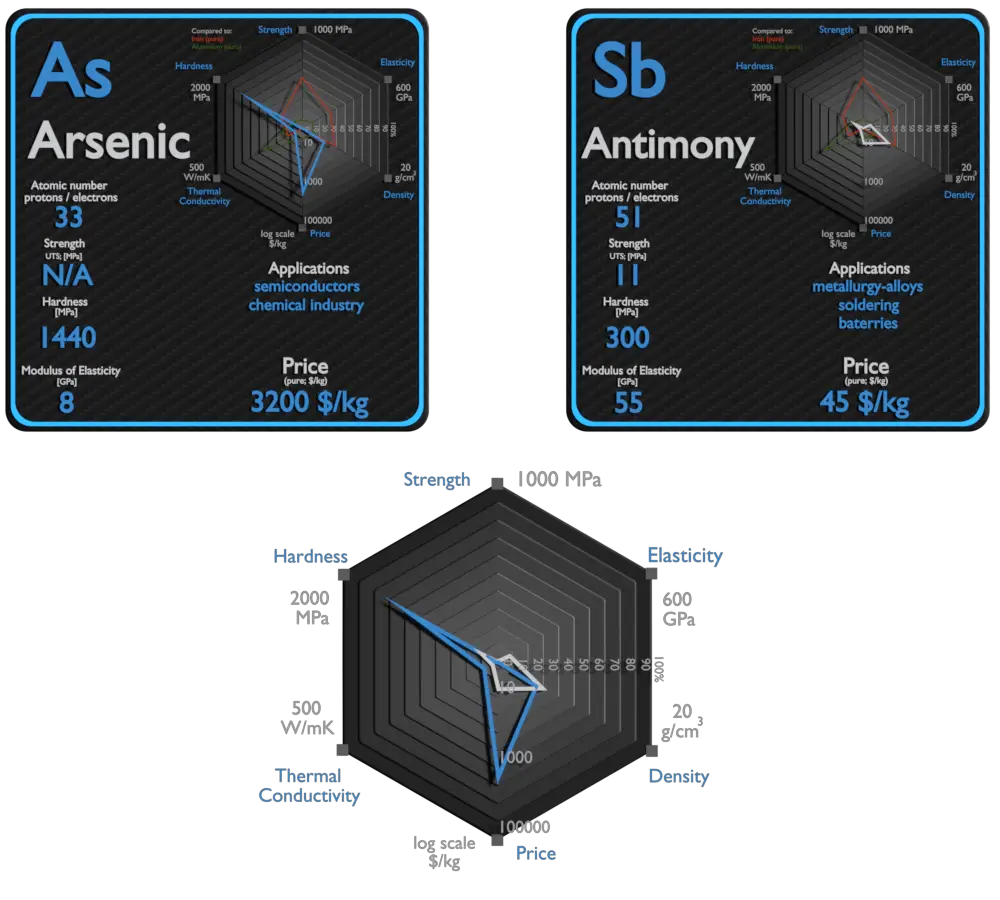
Arsenic and Antimony – About Elements


Source: www.luciteria.com
Arsenic and Antimony – Applications
Arsenic
Arsenic is used as a doping agent in semiconductors (gallium arsenide) for solid-state devices. It is also used in bronzing, pyrotechnics and for hardening shot. Arsenic compounds can be used to make special glass and preserve wood.
Antimony
The largest applications for metallic antimony are an alloy with lead and tin and the lead antimony plates in lead–acid batteries. Alloys of lead and tin with antimony have improved properties for solders, bullets, and plain bearings. Antimony can be used in fire retardants for many commercial and domestic products. Antimony trichloride is used in the manufacturing flame-proofing compounds as well as paints, ceramic enamels, glass and pottery. Other uses include ball bearings and mixing with alloys with percentages ranging from 1 to 20 greatly increasing the hardness and mechanical strength of the lead. The capability to strengthen already strong alloys is its largest and most widespread use.
Arsenic and Antimony – Comparison in Table
| Element | Arsenic | Antimony |
| Density | 5.727 g/cm3 | 6.697 g/cm3 |
| Ultimate Tensile Strength | N/A | 11 MPa |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | 8 GPa | 55 GPa |
| Mohs Scale | 3.5 | 3.15 |
| Brinell Hardness | 1440 MPa | 300 MPa |
| Vickers Hardness | N/A | N/A |
| Melting Point | 817 °C | 631 °C |
| Boiling Point | 614 °C | 1950 °C |
| Thermal Conductivity | 50 W/mK | 24 W/mK |
| Thermal Expansion Coefficient | 5.6 µm/mK | 11 µm/mK |
| Specific Heat | 0.33 J/g K | 0.21 J/g K |
| Heat of Fusion | N/A | 19.87 kJ/mol |
| Heat of Vaporization | 34.76 kJ/mol | 77.14 kJ/mol |





