This article contains comparison of key thermal and atomic properties of rhodium and rhenium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Rhodium vs Rhenium.
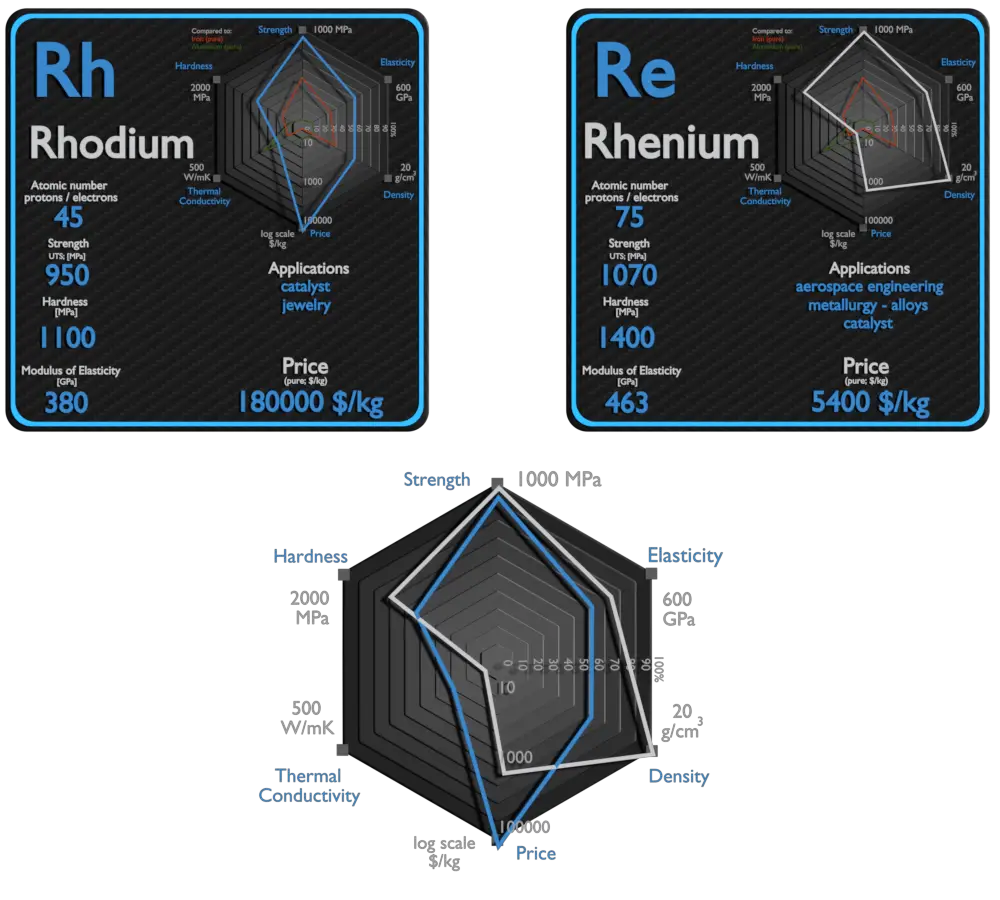
Rhodium and Rhenium – About Elements


Source: www.luciteria.com
Rhodium and Rhenium – Applications
Rhodium
The element’s major use (approximately 80% of world rhodium production) is as one of the catalysts in the three-way catalytic converters in automobiles. Because rhodium metal is inert against corrosion and most aggressive chemicals, and because of its rarity, rhodium is usually alloyed with platinum or palladium and applied in high-temperature and corrosion-resistive coatings. In nuclear reactors, rhodium-based detectors are often used for incore neutron flux measuring.
Rhenium
More than 80% of the global rhenium usage is in high-temperature superalloys for aircraft applications like turbine blades and engine parts. These alloys contain up to 6% rhenium, making jet engine construction the largest single use for the element. Rhenium is added to nickel-based super alloys to improve creep strength of the alloys. The remaining demand is mostly from petrochemical refining industries. Platinum-rhenium catalysts that are chiefly used in lead-free, high-octane gasoline are another major application of rhenium.
Rhodium and Rhenium – Comparison in Table
| Element | Rhodium | Rhenium |
| Density | 12.45 g/cm3 | 21.02 g/cm3 |
| Ultimate Tensile Strength | 950 MPa | 1070 MPa |
| Yield Strength | N/A | 290 MPa |
| Young’s Modulus of Elasticity | 380 GPa | 463 GPa |
| Mohs Scale | 6 | 7 |
| Brinell Hardness | 1100 MPa | 1400 MPa |
| Vickers Hardness | 1246 MPa | 2500 MPa |
| Melting Point | 1964 °C | 3180 °C |
| Boiling Point | 3695 °C | 5600 °C |
| Thermal Conductivity | 150 W/mK | 48 W/mK |
| Thermal Expansion Coefficient | 8.2 µm/mK | 6.2 µm/mK |
| Specific Heat | 0.242 J/g K | 0.13 J/g K |
| Heat of Fusion | 21.5 kJ/mol | 33.2 kJ/mol |
| Heat of Vaporization | 493 kJ/mol | 715 kJ/mol |







