This article contains comparison of key thermal and atomic properties of strontium and iodine, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Strontium vs Iodine.
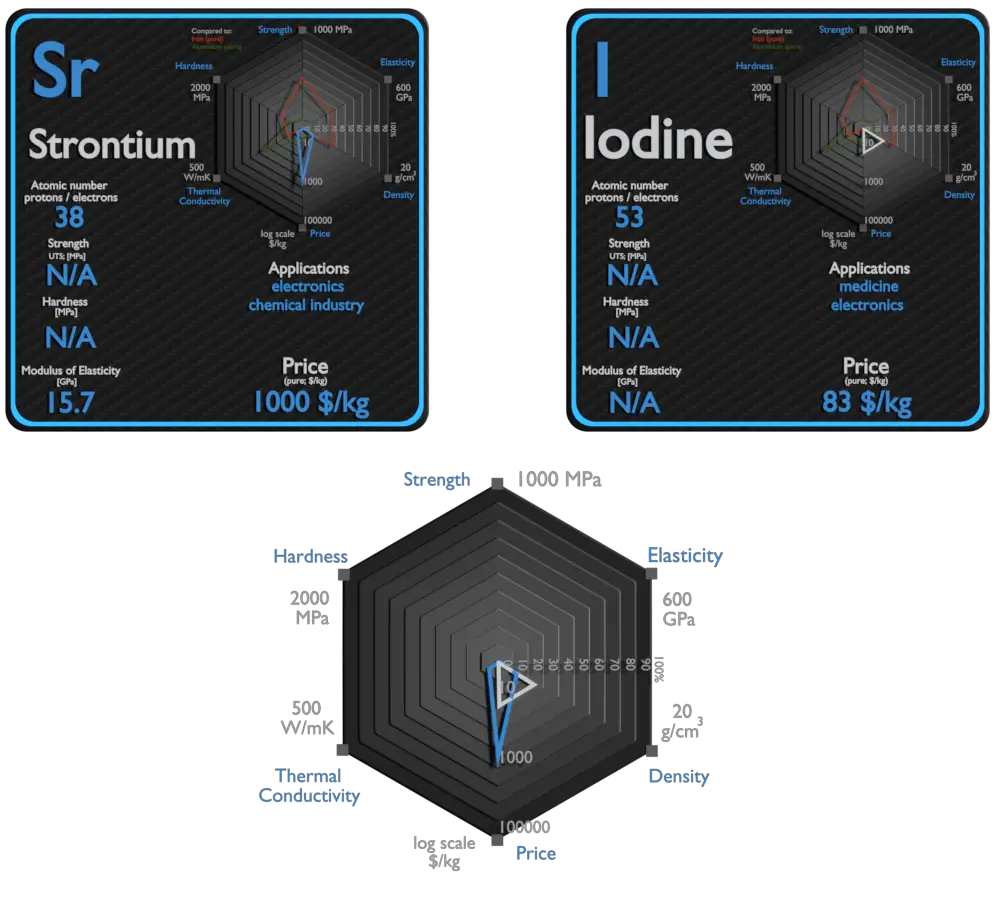
Strontium and Iodine – About Elements


Source: www.luciteria.com
Strontium and Iodine – Applications
Strontium
Consuming 75% of production, the primary use for strontium was in glass for colour television cathode ray tubes. Strontium is best known for the brilliant reds its salts give to fireworks and flares. It is also used in producing ferrite magnets and refining zinc. Modern ‘glow-in-the-dark’ paints and plastics contain strontium aluminate. They absorb light during the day and release it slowly for hours afterwards. The isotope Sr90 has a half-life of 28 years and is one of the well-known high-energy beta emitters. Hence, it is employed in systems for nuclear auxiliary power (SNAP) devices, which find potential applications in remote weather stations, space vehicles, navigational buoys, etc.
Iodine
In addition to nutrition products, iodine and iodine derivatives are used in a wide range of medical, agricultural, and industrial applications. About half of all produced iodine goes into various organoiodine compounds, another 15% remains as the pure element, another 15% is used to form potassium iodide, and another 15% for other inorganic iodine compounds. The leading application is in the production of X-ray contrast media (22%). Iodine’s high atomic number and density make it ideally suited for this application, as its presence in the body can help to increase contrast between tissues, organs, and blood vessels with similar X-ray densities. It is used as an antiseptic for external wounds. Another application driving the demand for iodine is in polarizing film in liquidcrystal display (LCD) screens.
Strontium and Iodine – Comparison in Table
| Element | Strontium | Iodine |
| Density | 2.63 g/cm3 | 4.94 g/cm3 |
| Ultimate Tensile Strength | N/A | N/A |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | 15.7 GPa | N/A |
| Mohs Scale | 1.8 | N/A |
| Brinell Hardness | N/A | N/A |
| Vickers Hardness | N/A | N/A |
| Melting Point | 777 °C | 113.5 °C |
| Boiling Point | 1382 °C | 184 °C |
| Thermal Conductivity | 35.3 W/mK | 0.449 W/mK |
| Thermal Expansion Coefficient | 22.5 µm/mK | N/A |
| Specific Heat | 0.3 J/g K | 0.214 J/g K |
| Heat of Fusion | 8.3 kJ/mol | 7.824 kJ/mol |
| Heat of Vaporization | 144 kJ/mol | 20.752 kJ/mol |







