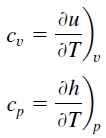About Iodine
Iodine is the heaviest of the stable halogens, it exists as a lustrous, purple-black metallic solid at standard conditions that sublimes readily to form a violet gas. Iodine is the least abundant of the stable halogens, being the sixty-first most abundant element. It is even less abundant than the so-called rare earths. It is the heaviest essential mineral nutrient.
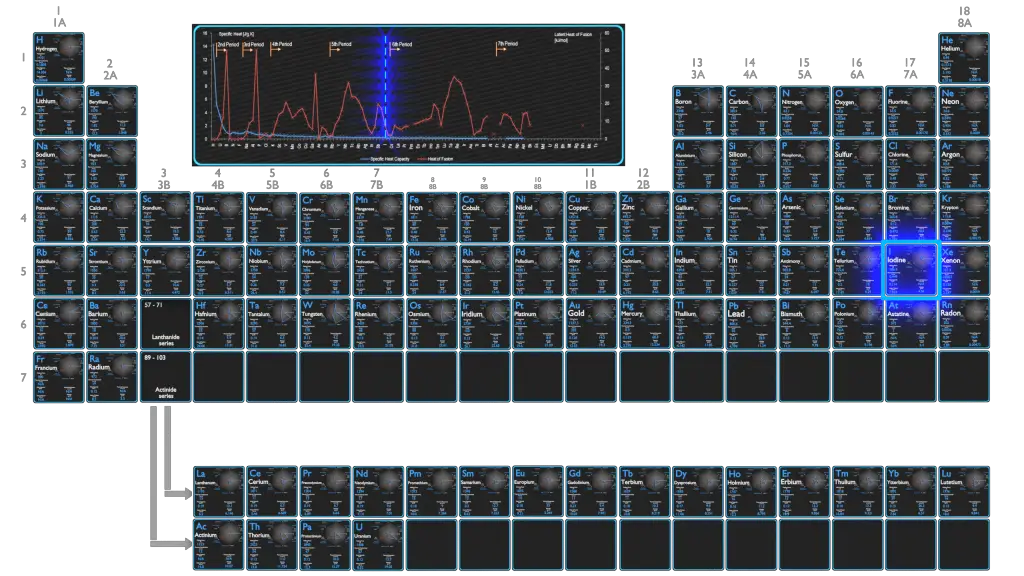
Iodine – Specific Heat, Latent Heat of Fusion, Latent Heat of Vaporization
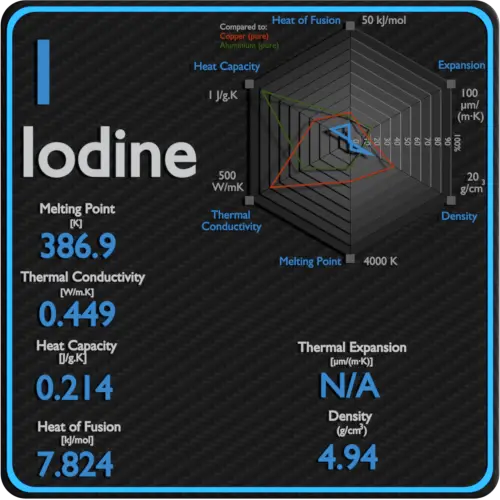
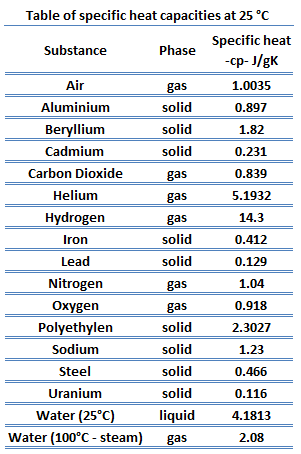
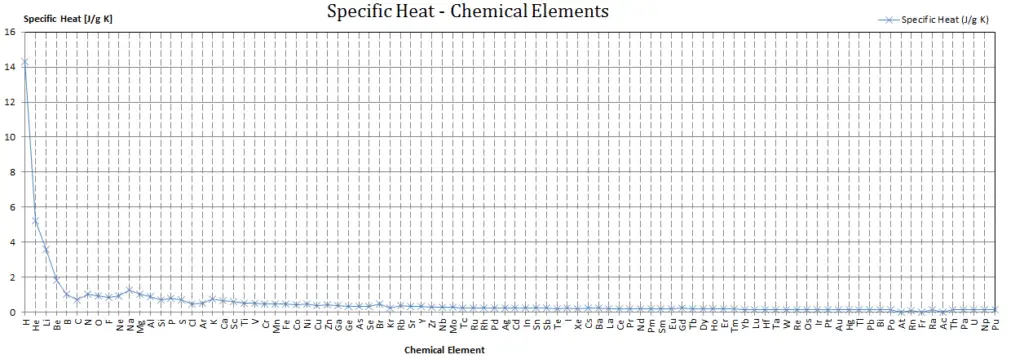
Specific heat of Iodine is 0.214 J/g K.
Heat capacity is an extensive property of matter, meaning it is proportional to the size of the system. Heat capacity C has the unit of energy per degree or energy per kelvin. When expressing the same phenomenon as an intensive property, the heat capacity is divided by the amount of substance, mass, or volume, thus the quantity is independent of the size or extent of the sample.
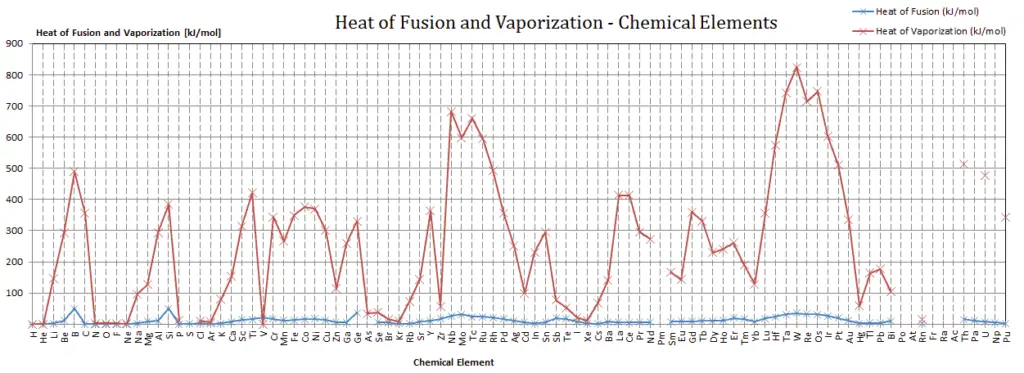
Latent Heat of Fusion of Iodine is 7.824 kJ/mol.
Latent Heat of Vaporization of Iodine is 20.752 kJ/mol.
Latent heat is the amount of heat added to or removed from a substance to produce a change in phase. This energy breaks down the intermolecular attractive forces, and also must provide the energy necessary to expand the gas (the pΔV work). When latent heat is added, no temperature change occurs. The enthalpy of vaporization is a function of the pressure at which that transformation takes place.
See also: Mechanical Properties of Iodine
Summary
| Element | Iodine |
| Specific Heat | 0.214 J/g K |
| Heat of Fusion | 7.824 kJ/mol |
| Heat of Vaporization | 20.752 kJ/mol |
| Density | 4.94 g/cm3 |
Source: www.luciteria.com