This article contains comparison of key thermal and atomic properties of boron and silicon, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Boron vs Silicon.
![]()
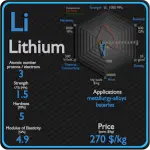
Boron and Silicon – About Elements

![]()
Source: www.luciteria.com
Boron and Silicon – Applications
Boron
Nearly all boron ore extracted from the Earth is destined for refinement into boric acid and sodium tetraborate pentahydrate. In the United States, 70% of the boron is used for the production of glass and ceramics. The major global industrial-scale use of boron compounds (about 46% of end-use) is in production of glass fiber for boron-containing insulating and structural fiberglasses, especially in Asia. Boron is added to boron steels at the level of a few parts per million to increase hardenability. Higher percentages are added to steels used in the nuclear industry due to boron’s neutron absorption ability (e.g. pellets of Boron Carbide). Boron can also increase the surface hardness of steels and alloys through boriding. Boron carbide and cubic boron nitride powders are widely used as abrasives.
Silicon
Most silicon is used industrially without being purified, and indeed, often with comparatively little processing from its natural form. Silicon is a vital ingredient in aluminum, steel, and iron alloys. It is added as a fluxing agent for copper alloys. In the form of clay and sand, it is used to manufacture bricks and concrete; it is a valuable refractory material for high-temperature work, for example, molding sands for castings in foundry applications. Silica is used to make fire brick, a type of ceramic. Silicate minerals are also in whiteware ceramics, an important class of products usually containing various types of fired clay minerals (natural aluminium phyllosilicates). An example is porcelain, which is based on the silicate mineral kaolinite. Traditional glass (silica-based soda-lime glass) also functions in many of the same ways, and also is used for windows and containers. Hyperpure silicon metal and doped hyperpure silicon (doping with boron, phosphorous, gallium, or arsenic) are used in solar cells, transistors and semiconductors.
Boron and Silicon – Comparison in Table
| Element | Boron | Silicon |
| Density | 2.46 g/cm3 | 2.33 g/cm3 |
| Ultimate Tensile Strength | N/A | 170 MPa |
| Yield Strength | N/A | 165 MPa |
| Young’s Modulus of Elasticity | N/A | 150 GPa |
| Mohs Scale | 9.5 | 7 |
| Brinell Hardness | N/A | N/A |
| Vickers Hardness | 49000 MPa | N/A |
| Melting Point | 2079 °C | 1410 °C |
| Boiling Point | 3927 °C | 3265 °C |
| Thermal Conductivity | 27 W/mK | 148 W/mK |
| Thermal Expansion Coefficient | 5-7 µm/mK | 2.6 µm/mK |
| Specific Heat | 1.02 J/g K | 0.71 J/g K |
| Heat of Fusion | 50.2 kJ/mol | 50.55 kJ/mol |
| Heat of Vaporization | 508 kJ/mol | 384.22 kJ/mol |