This article contains comparison of key thermal and atomic properties of boron and carbon, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Boron vs Carbon.
Boron and Carbon – About Elements


Source: www.luciteria.com
Boron and Carbon – Applications
Boron
Nearly all boron ore extracted from the Earth is destined for refinement into boric acid and sodium tetraborate pentahydrate. In the United States, 70% of the boron is used for the production of glass and ceramics. The major global industrial-scale use of boron compounds (about 46% of end-use) is in production of glass fiber for boron-containing insulating and structural fiberglasses, especially in Asia. Boron is added to boron steels at the level of a few parts per million to increase hardenability. Higher percentages are added to steels used in the nuclear industry due to boron’s neutron absorption ability (e.g. pellets of Boron Carbide). Boron can also increase the surface hardness of steels and alloys through boriding. Boron carbide and cubic boron nitride powders are widely used as abrasives.
Carbon
The major economic use of carbon other than food and wood is in the form of hydrocarbons, most notably the fossil fuel methane gas and crude oil (petroleum). Graphite and diamonds are two important allotropes of carbon that have wide applications. The uses of carbon and its compounds are extremely varied. It can form alloys with iron, of which the most common is carbon steel. Carbon is a non-metallic element, which is an important alloying element in all ferrous metal based materials. Carbon is always present in metallic alloys, i.e. in all grades of stainless steel and heat resistant alloys. Carbon is a very strong austenitizer and increases the strength of steel. In fact, it is the principal hardening element and is essential to the formation of cementite, Fe3C, pearlite, spheroidite, and iron-carbon martensite. Adding a small amount of non-metallic carbon to iron trades its great ductility for the greater strength. Graphite is combined with clays to form the ‘lead’ used in pencils used for writing and drawing. It is also used as a lubricant and a pigment, as a molding material in glass manufacture, in electrodes for dry batteries and in electroplating and electroforming, in brushes for electric motors and as a neutron moderator in nuclear reactors. Charcoal has been used since earliest times for a large range of purposes including art and medicine, but by far its most important use has been as a metallurgical fuel. Carbon fibers are used where low weight, high stiffness, high conductivity, or where the look of the carbon fiber weave desired.
Boron and Carbon – Comparison in Table
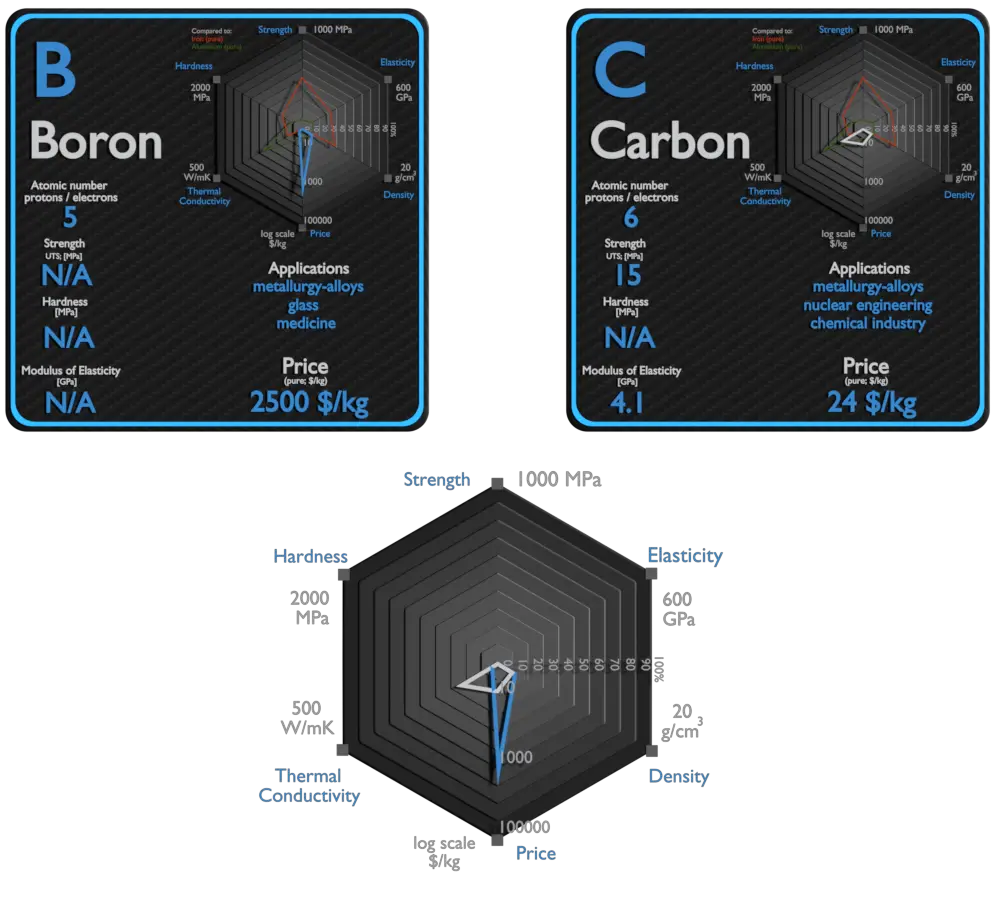
| Element | Boron | Carbon |
| Density | 2.46 g/cm3 | 2.26 g/cm3 |
| Ultimate Tensile Strength | N/A | 15 MPa (graphite); 3500 MPa (carbon fiber) |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | N/A | 4.1 GPa (graphite); 228 GPa (carbon fiber) |
| Mohs Scale | 9.5 | 0.8 (graphite) |
| Brinell Hardness | N/A | N/A |
| Vickers Hardness | 49000 MPa | N/A |
| Melting Point | 2079 °C | 4099 °C |
| Boiling Point | 3927 °C | 4527 °C |
| Thermal Conductivity | 27 W/mK | 129 W/mK |
| Thermal Expansion Coefficient | 5-7 µm/mK | 0.8 µm/mK |
| Specific Heat | 1.02 J/g K | 0.71 J/g K |
| Heat of Fusion | 50.2 kJ/mol | N/A |
| Heat of Vaporization | 508 kJ/mol | 355.8 kJ/mol |








