This article contains comparison of key thermal and atomic properties of iodine and caesium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Iodine vs Caesium.
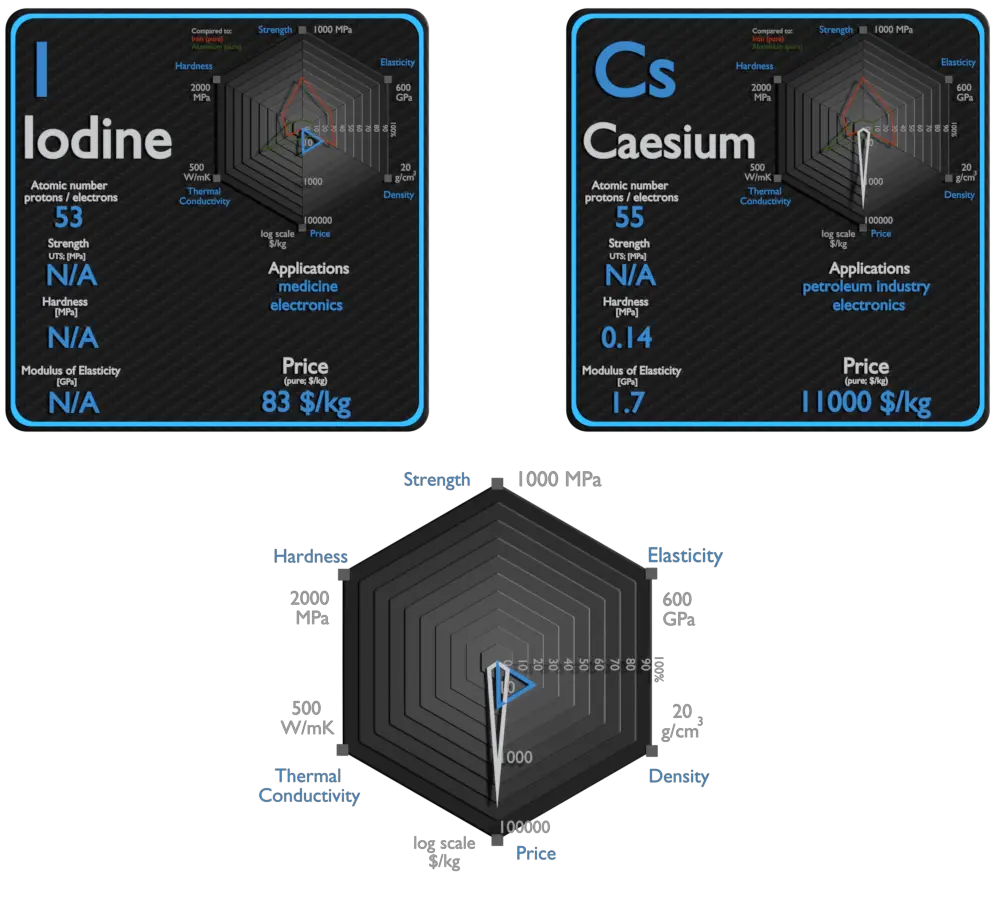
Iodine and Caesium – About Elements


Source: www.luciteria.com
Iodine and Caesium – Applications
Iodine
In addition to nutrition products, iodine and iodine derivatives are used in a wide range of medical, agricultural, and industrial applications. About half of all produced iodine goes into various organoiodine compounds, another 15% remains as the pure element, another 15% is used to form potassium iodide, and another 15% for other inorganic iodine compounds. The leading application is in the production of X-ray contrast media (22%). Iodine’s high atomic number and density make it ideally suited for this application, as its presence in the body can help to increase contrast between tissues, organs, and blood vessels with similar X-ray densities. It is used as an antiseptic for external wounds. Another application driving the demand for iodine is in polarizing film in liquidcrystal display (LCD) screens.
Caesium
The largest present-day use of nonradioactive caesium is in caesium formate drilling fluids for the extractive oil industry. They are also used to make special optical glass, as a catalyst promoter, in vacuum tubes and in radiation monitoring equipment. One of its most important uses is in the ‘caesium clock’ (atomic clock). These clocks are a vital part of the internetand mobile phone networks, as well as Global Positioning System (GPS) satellites. Caesium-137 is a radioisotope commonly used as a gamma-emitter in industrial applications.
Iodine and Caesium – Comparison in Table
| Element | Iodine | Caesium |
| Density | 4.94 g/cm | 1.879 g/cm3 |
| Ultimate Tensile Strength | N/A | N/A |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | N/A | 1.7 GPa |
| Mohs Scale | N/A | 0.2 |
| Brinell Hardness | N/A | 0.14 MPa |
| Vickers Hardness | N/A | N/A |
| Melting Point | 113.5 °C | 28.4 °C |
| Boiling Point | 184 °C | 669 °C |
| Thermal Conductivity | 0.449 W/mK | 36 W/mK |
| Thermal Expansion Coefficient | N/A | 97 µm/mK |
| Specific Heat | 0.214 J/g K | 0.24 J/g K |
| Heat of Fusion | 7.824 kJ/mol | 2.092 kJ/mol |
| Heat of Vaporization | 20.752 kJ/mol | 67.74 kJ/mol |







