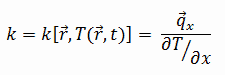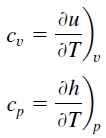About Propane
Propane is a colourless, easily liquefied, gaseous hydrocarbon (alkane). The chemical formula for propane is C3H8. It is separated in large quantities from natural gas, light crude oil, and oil-refinery gases and is commercially available as liquefied propane or as a major constituent of liquefied petroleum gas (LPG). Propane gas has become a popular choice for barbecues and portable stoves because its low boiling point makes it vaporize as soon as it is released from its pressurized container. At normal pressure it liquifies below its boiling point at −42 °C and solidifies below its melting point at −187.7 °C.
Summary
| Name | Propane |
| Phase at STP | gas |
| Density | 2 kg/m3 |
| Ultimate Tensile Strength | N/A |
| Yield Strength | N/A |
| Young’s Modulus of Elasticity | N/A |
| Brinell Hardness | N/A |
| Melting Point | -189 °C |
| Thermal Conductivity | 0.017 W/mK |
| Heat Capacity | 1630 J/g K |
| Price | 1.5 $/kg |
Composition of Propane
Propane is a three-carbon alkane with the molecular formula C3H8.
Applications of Propane

Propane is commonly used as a fuel in domestic and industrial applications and in low-emissions public.
Thermal Properties of Propane
Propane – Melting Point
Melting point of Propane is 1-189 °C.
Note that, these points are associated with the standard atmospheric pressure. In general, melting is a phase change of a substance from the solid to the liquid phase. The melting point of a substance is the temperature at which this phase change occurs. The melting point also defines a condition in which the solid and liquid can exist in equilibrium. For various chemical compounds and alloys, it is difficult to define the melting point, since they are usually a mixture of various chemical elements.
Propane – Thermal Conductivity
Thermal conductivity of Propane is 0.017 W/(m·K).
The heat transfer characteristics of a solid material are measured by a property called the thermal conductivity, k (or λ), measured in W/m.K. It is a measure of a substance’s ability to transfer heat through a material by conduction. Note that Fourier’s law applies for all matter, regardless of its state (solid, liquid, or gas), therefore, it is also defined for liquids and gases.
The thermal conductivity of most liquids and solids varies with temperature. For vapors, it also depends upon pressure. In general:
Most materials are very nearly homogeneous, therefore we can usually write k = k (T). Similar definitions are associated with thermal conductivities in the y- and z-directions (ky, kz), but for an isotropic material the thermal conductivity is independent of the direction of transfer, kx = ky = kz = k.
Propane – Specific Heat
Specific heat of Propane is 1630 J/g K.
Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The intensive properties cv and cp are defined for pure, simple compressible substances as partial derivatives of the internal energy u(T, v) and enthalpy h(T, p), respectively:
where the subscripts v and p denote the variables held fixed during differentiation. The properties cv and cp are referred to as specific heats (or heat capacities) because under certain special conditions they relate the temperature change of a system to the amount of energy added by heat transfer. Their SI units are J/kg K or J/mol K.
Properties and prices of other materials
material-table-in-8k-resolution