This article contains comparison of key thermal and atomic properties of sodium and calcium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Sodium vs Calcium.
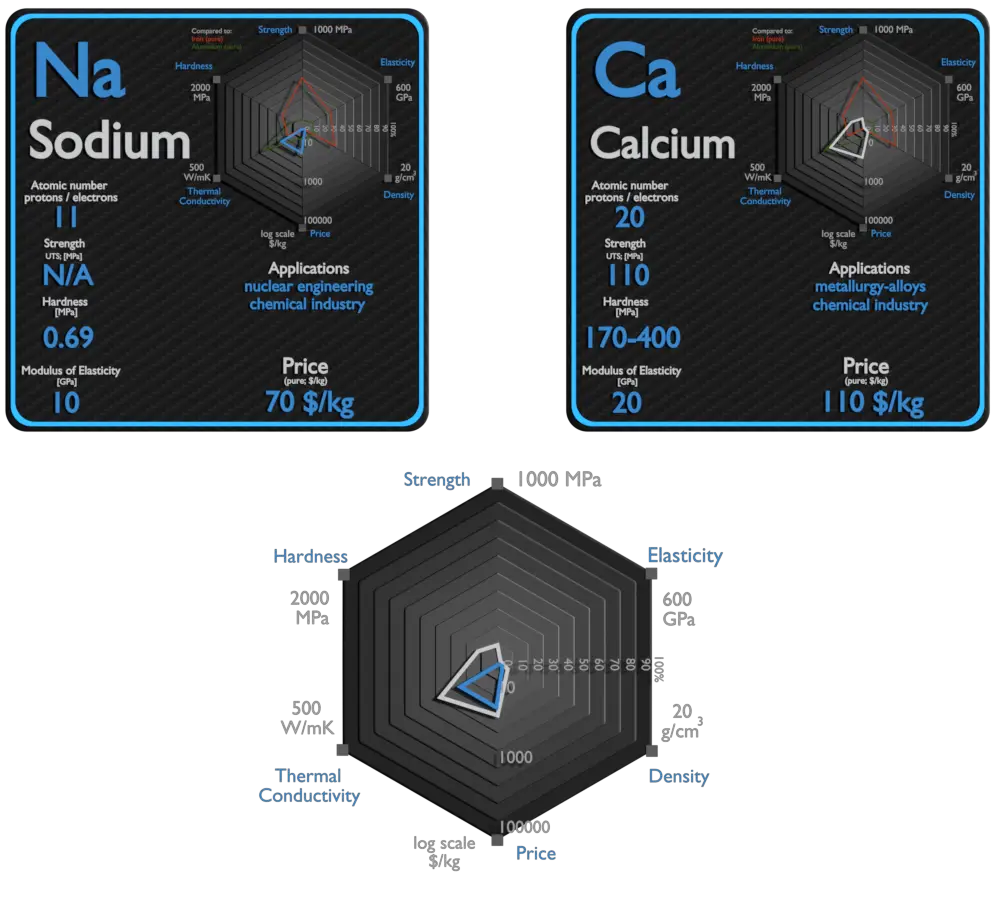
Sodium and Calcium – About Elements


Source: www.luciteria.com
Sodium and Calcium – Applications
Sodium
Metallic sodium is used mainly for the production of sodium borohydride, sodium azide, indigo, and triphenylphosphine. A once-common use was the making of tetraethyllead and titanium metal; because of the move away from TEL and new titanium production methods. An electric current and sodium vapor combine to form a yellowish glow. This principle is used for the making of sodium vapor lamps. Sodium is occasionally used as a heat exchange medium in nuclear power plants. Liquid sodium is sealed into pipes surrounding the reactor core. Generated heat is absorbed by sodium and forced through the pipes in a heat exchanger which can be used to generate electricity.
Calcium
The largest use of metallic calcium is in steelmaking, due to its strong chemical affinity for oxygen and sulfur. Its oxides and sulfides, once formed, give liquid lime aluminate and sulfide inclusions in steel which float out. Calcium compounds are used as manufacture of insecticides, paints, blackboard chalk, textile and fireworks.
Sodium and Calcium – Comparison in Table
| Element | Sodium | Calcium |
| Density | 0.968 g/cm3 | 1.55 g/cm3 |
| Ultimate Tensile Strength | N/A | 110 MPa |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | 10 GPa | 20 GPa |
| Mohs Scale | 0.4 | 1.5 |
| Brinell Hardness | 0.69 MPa | 170 – 400 MPa |
| Vickers Hardness | N/A | N/A |
| Melting Point | 97.8 °C | 842 °C |
| Boiling Point | 883 °C | 1484 °C |
| Thermal Conductivity | 141 W/mK | 200 W/mK |
| Thermal Expansion Coefficient | 71 µm/mK | 22.3 µm/mK |
| Specific Heat | 1.23 J/g K | 0.63 J/g K |
| Heat of Fusion | 2.598 kJ/mol | 8.54 kJ/mol |
| Heat of Vaporization | 96.96 kJ/mol | 153.3 kJ/mol |