This article contains comparison of key thermal and atomic properties of calcium and iron, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Calcium vs Iron.
Calcium and Iron – About Elements


Source: www.luciteria.com
Calcium and Iron – Applications
Calcium
The largest use of metallic calcium is in steelmaking, due to its strong chemical affinity for oxygen and sulfur. Its oxides and sulfides, once formed, give liquid lime aluminate and sulfide inclusions in steel which float out. Calcium compounds are used as manufacture of insecticides, paints, blackboard chalk, textile and fireworks.
Iron
Iron is used in numerous sectors such as electronics, manufacturing, automotive, and construction and building. Iron is the most widely used of all the metals, accounting for over 90% of worldwide metal produc0tion. Its low cost and high strength often make it the material of choice material to withstand stress or transmit forces, such as the construction of machinery and machine tools, rails, automobiles, ship hulls, concrete reinforcing bars, and the load-carrying framework of buildings. Since pure iron is quite soft, it is most commonly combined with alloying elements to make steel. Steels are iron–carbon alloys that may contain appreciable concentrations of other alloying elements. Adding a small amount of non-metallic carbon to iron trades its great ductility for the greater strength. Due to its very-high strength, but still substantial toughness, and its ability to be greatly altered by heat treatment, steel is one of the most useful and common ferrous alloy in modern use. There are thousands of alloys that have different compositions and/or heat treatments. The mechanical properties are sensitive to the content of carbon, which is normally less than 1.0 wt%.
Calcium and Iron – Comparison in Table
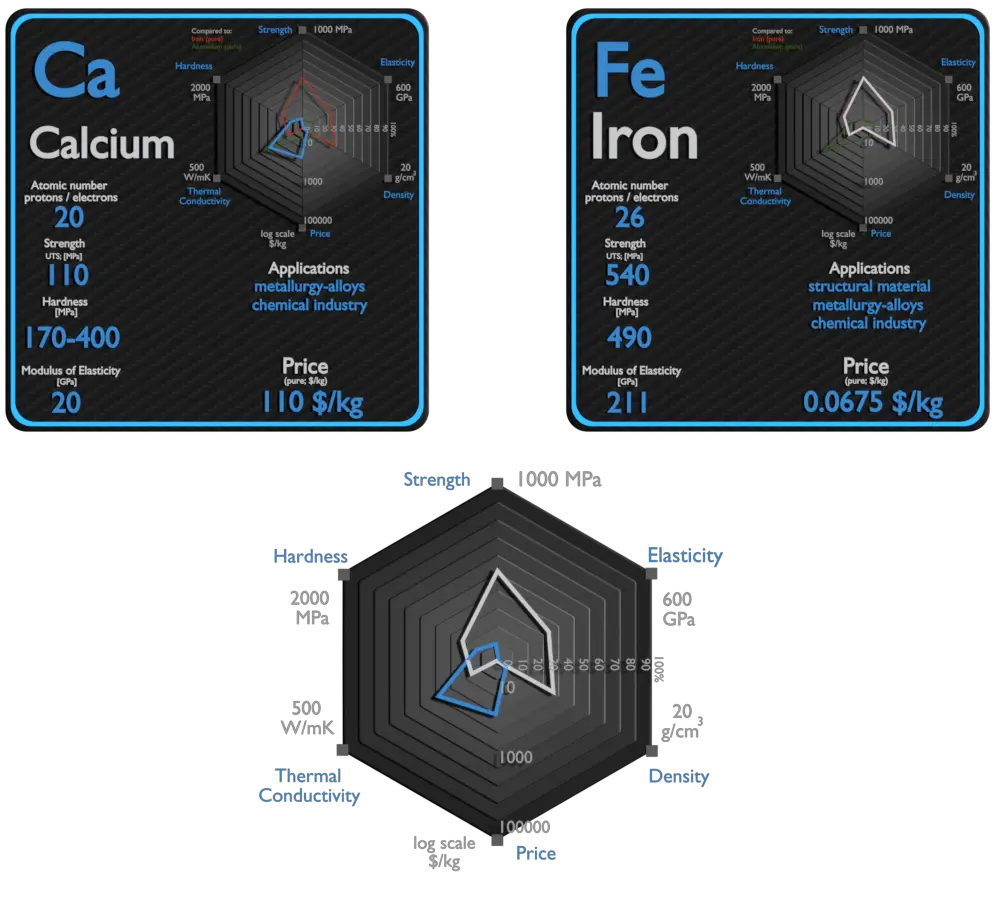
| Element | Calcium | Iron |
| Density | 1.55 g/cm3 | 7.874 g/cm3 |
| Ultimate Tensile Strength | 110 MPa | 540 MPa |
| Yield Strength | N/A | 50 MPa |
| Young’s Modulus of Elasticity | 20 GPa | 211 GPa |
| Mohs Scale | 1.5 | 4.5 |
| Brinell Hardness | 170 – 400 MPa | 490 MPa |
| Vickers Hardness | N/A | 608 MPa |
| Melting Point | 842 °C | 1538 °C |
| Boiling Point | 1484 °C | 2861 °C |
| Thermal Conductivity | 200 W/mK | 80.2 W/mK |
| Thermal Expansion Coefficient | 22.3 µm/mK | 11.8 µm/mK |
| Specific Heat | 0.63 J/g K | 0.44 J/g K |
| Heat of Fusion | 8.54 kJ/mol | 13.8 kJ/mol |
| Heat of Vaporization | 153.3 kJ/mol | 349.6 kJ/mol |