This article contains comparison of key thermal and atomic properties of titanium and niobium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Titanium vs Niobium.
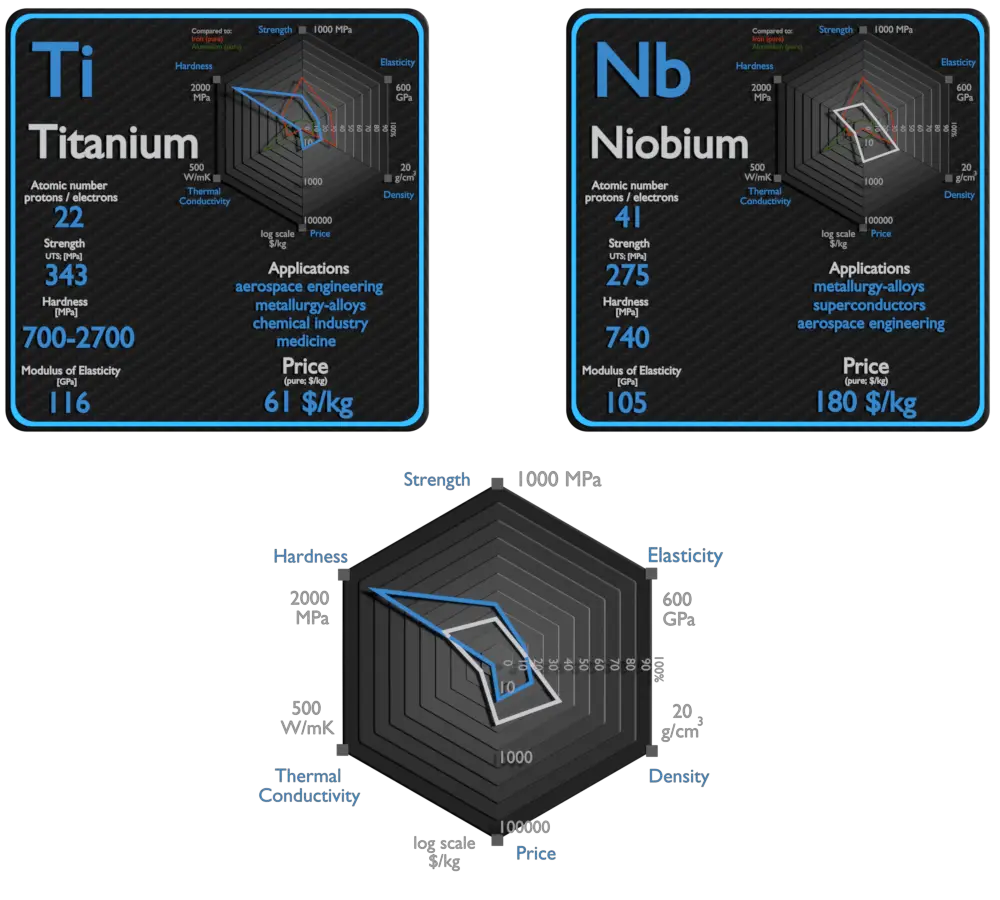
Titanium and Niobium – About Elements


Source: www.luciteria.com
Titanium and Niobium – Applications
Titanium
The two most useful properties of the metal are corrosion resistance and strength-to-density ratio, the highest of any metallic element. The corrosion resistance of titanium alloys at normal temperatures is unusually high. These properties determine application of titanium and its alloys. The earliest production application of titanium was in 1952, for the nacelles and firewalls of the Douglas DC-7 airliner. High specific strength, good fatigue resistance and creep life, and good fracture toughness are characteristics that make titanium a preferred metal for aerospace applications. Aerospace applications, including use in both structural (airframe) components and jet engines, still account for the largest share of titanium alloy use. On the supersonic aircraft SR-71, titanium was used for 85% of the structure. Due to very high inertness, titanium has many biomedical applications, which is based on its inertness in the human body, that is, resistance to corrosion by body fluids.
Niobium
Niobium consumption is dominated by its use as additive to high strength low alloy steel and stainless steel for oil and gas pipelines, car and truck bodies, architectural requirements, tool steels, ships hulls, railroad tracks. However, there are a number of other applications for niobium metal and its compounds. Although niobium has many applications the majority is used in the production of high-grade structural steel. The second largest application for niobium is in nickel-based superalloys. Niobium-tin alloys are used as superconducting magnets.
Titanium and Niobium – Comparison in Table
| Element | Titanium | Niobium |
| Density | 4.507 g/cm3 | 8.57 g/cm3 |
| Ultimate Tensile Strength | 434 MPa, 293 MPa (pure) | 275 MPa |
| Yield Strength | 380 MPa | 70 MPa |
| Young’s Modulus of Elasticity | 116 GPa | 105 GPa |
| Mohs Scale | 6 | 6 |
| Brinell Hardness | 700 – 2700 MPa | 740 MPa |
| Vickers Hardness | 800 – 3400 MPa | 1300 MPa |
| Melting Point | 1668 °C | 2477 °C |
| Boiling Point | 3287 °C | 4744 °C |
| Thermal Conductivity | 21.9 W/mK | 53.7 W/mK |
| Thermal Expansion Coefficient | 8.6 µm/mK | 7.3 µm/mK |
| Specific Heat | 0.52 J/g K | 0.26 J/g K |
| Heat of Fusion | 15.45 kJ/mol | 26.4 kJ/mol |
| Heat of Vaporization | 421 kJ/mol | 682 kJ/mol |











