This article contains comparison of key thermal and atomic properties of lithium and nitrogen, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Lithium vs Nitrogen.

Lithium and Nitrogen – About Elements


Source: www.luciteria.com
Lithium and Nitrogen – Applications
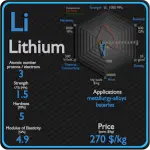
Lithium
Lithium has many applications, from lubricating grease, alloying additions in particular for aluminium and magnesium alloys, to glazes for ceramics, and finally, lithium batteries. In particular, lithium is and will continue to play an increasingly important role in the battery-powered clean air future. Lithium batteries are widely used in portable consumer electronic devices, and in electric vehicles ranging from full sized vehicles to radio controlled toys. The term “lithium battery” refers to a family of different lithium-metal chemistries, comprising many types of cathodes and electrolytes but all with metallic lithium as the anode.
Nitrogen
Nitrogen in various chemical forms plays a major role in large number of environmental issues. The applications of nitrogen compounds are naturally extremely widely varied due to the huge size of this class: hence, only applications of pure nitrogen itself will be considered here. Two-thirds of nitrogen produced by industry is sold as the gas and the remaining one-third as the liquid. In metallurgy, nitriding is a case hardening process in which the surface nitrogen concentration of a ferrous is increased by diffusion from the surrounding environment to create case-hardened surface. Nitriding produces hard, highly wear-resistant surface (shallow case depths) of product with fair capacity for contact load, good bending fatigue strength and excellent resistance to seizure. Synthetically produced ammonia and nitrates are key industrial fertilisers, and fertiliser nitrates are key pollutants in the eutrophication of water systems. Apart from its use in fertilisers and energy-stores, nitrogen is a constituent of organic compounds as diverse as Kevlar used in high-strength fabric and cyanoacrylate used in superglue.
Lithium and Nitrogen – Comparison in Table
| Element | Lithium | Nitrogen |
| Density | 0.535 g/cm3 | 0.00125 g/cm3 |
| Ultimate Tensile Strength | 1.5 MPa | N/A |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | 4.9 GPa | N/A |
| Mohs Scale | 0.6 | N/A |
| Brinell Hardness | 5 MPa | N/A |
| Vickers Hardness | N/A | N/A |
| Melting Point | 180.5 °C | -209.9 °C |
| Boiling Point | 1342 °C | -195.8 °C |
| Thermal Conductivity | 85 W/mK | 0.02598 W/mK |
| Thermal Expansion Coefficient | 46 µm/mK | — µm/mK |
| Specific Heat | 3.6 J/g K | 1.04 J/g K |
| Heat of Fusion | 3 kJ/mol | (N2) 0.7204 kJ/mol |
| Heat of Vaporization | 145.92 kJ/mol | (N2) 5.56 kJ/mol |