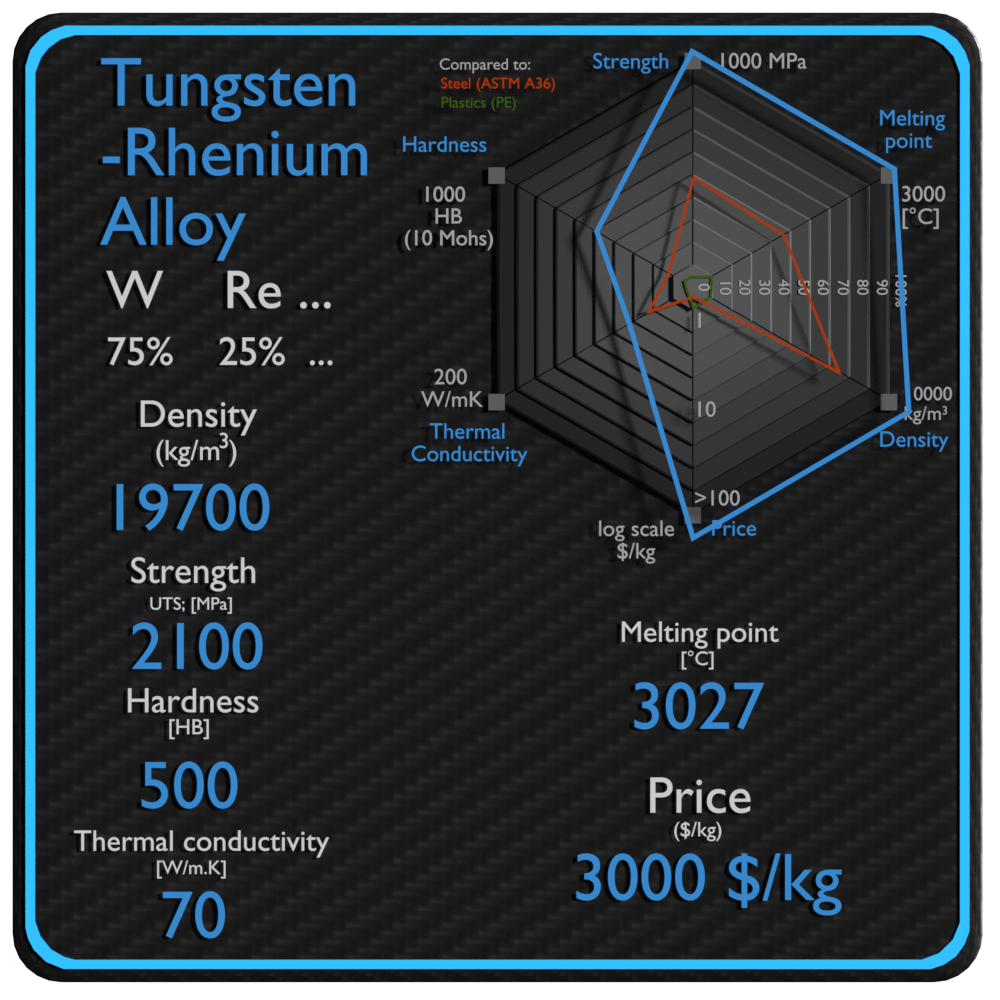
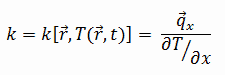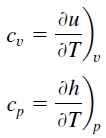About Tungsten-Rhenium Alloy
Tungsten and rhenium are both refractory metals. These metals are well known for their extraordinary resistance to heat and wear. Key requirement to withstand high temperatures is a high melting point and stable mechanical properties (e.g. high hardness) even at high temperatures. These metals are usually combined together to obtain desired fabricability, thermal and mechanical properties. Powder-metallurgy methods can be used in consolidating tungsten-rhenium alloys. Up to 22% rhenium is alloyed with tungsten to improve its high temperature strength and corrosion resistance. The hardness of as-cast W – 30Re alloy is around 500 BHN. This hardness strongly depends on rhenium content.
Summary
| Name | Tungsten-Rhenium Alloy |
| Phase at STP | solid |
| Density | 19700 kg/m3 |
| Ultimate Tensile Strength | 2100 MPa |
| Yield Strength | N/A |
| Young’s Modulus of Elasticity | 400 GPa |
| Brinell Hardness | 500 BHN |
| Melting Point | 3027 °C |
| Thermal Conductivity | 70 W/mK |
| Heat Capacity | 140 J/g K |
| Price | 3000 $/kg |
Composition of Tungsten-Rhenium Alloy
Tungsten-rhenium alloys typically contains 10, 20, 30, 35, and 40% of rhenium.
Applications of Tungsten-Rhenium Alloy
Historically Tungsten 25% Rhenium alloy has been produced into wire for the thermocouple market, but recent demands for high temperature structural components have forced the development of novel processing techniques for Tungsten-Rhenium and Tungsten-Rhenium with hafnium carbide. The hafnium carbide addition is known to increase strength of refractory metals. The applications for these materials are in the joining, medical, aerospace, furnace, and heat treat industries. Rhenium is considered highly desirable as an alloying addition with other refractory metals. Tungsten-rhenium alloys offer the highest temperature strength of any metal; the combination drastically increases ductility and tensile strength. Rhenium is gaining acceptance in nuclear reactors, miniature rockets, and other commercial and aerospace applications.
Mechanical Properties of Tungsten-Rhenium Alloy
Strength of Tungsten-Rhenium Alloy
In mechanics of materials, the strength of a material is its ability to withstand an applied load without failure or plastic deformation. Strength of materials basically considers the relationship between the external loads applied to a material and the resulting deformation or change in material dimensions. In designing structures and machines, it is important to consider these factors, in order that the material selected will have adequate strength to resist applied loads or forces and retain its original shape.
Strength of a material is its ability to withstand this applied load without failure or plastic deformation. For tensile stress, the capacity of a material or structure to withstand loads tending to elongate is known as ultimate tensile strength (UTS). Yield strength or yield stress is the material property defined as the stress at which a material begins to deform plastically whereas yield point is the point where nonlinear (elastic + plastic) deformation begins. In case of tensional stress of a uniform bar (stress-strain curve), the Hooke’s law describes behaviour of a bar in the elastic region. The Young’s modulus of elasticity is the elastic modulus for tensile and compressive stress in the linear elasticity regime of a uniaxial deformation and is usually assessed by tensile tests.
See also: Strength of Materials
Ultimate Tensile Strength of Tungsten-Rhenium Alloy
Ultimate tensile strength of Tungsten-Rhenium Alloy is 2100 MPa.
Yield Strength of Tungsten-Rhenium Alloy
Yield strength of Tungsten-Rhenium Alloy is N/A.
Modulus of Elasticity of Tungsten-Rhenium Alloy
The Young’s modulus of elasticity of Tungsten-Rhenium Alloy is 400 GPa.
Hardness of Tungsten-Rhenium Alloy
In materials science, hardness is the ability to withstand surface indentation (localized plastic deformation) and scratching. Brinell hardness test is one of indentation hardness tests, that has been developed for hardness testing. In Brinell tests, a hard, spherical indenter is forced under a specific load into the surface of the metal to be tested.
The Brinell hardness number (HB) is the load divided by the surface area of the indentation. The diameter of the impression is measured with a microscope with a superimposed scale. The Brinell hardness number is computed from the equation:
Brinell hardness of Tungsten-Rhenium Alloy is approximately 500 BHN (converted).
See also: Hardness of Materials
Thermal Properties of Tungsten-Rhenium Alloy
Tungsten-Rhenium Alloy – Melting Point
Melting point of Tungsten-Rhenium Alloy is 3027 °C.
Note that, these points are associated with the standard atmospheric pressure. In general, melting is a phase change of a substance from the solid to the liquid phase. The melting point of a substance is the temperature at which this phase change occurs. The melting point also defines a condition in which the solid and liquid can exist in equilibrium. For various chemical compounds and alloys, it is difficult to define the melting point, since they are usually a mixture of various chemical elements.
Tungsten-Rhenium Alloy – Thermal Conductivity
Thermal conductivity of Tungsten-Rhenium Alloy is 70 W/(m·K).
The heat transfer characteristics of a solid material are measured by a property called the thermal conductivity, k (or λ), measured in W/m.K. It is a measure of a substance’s ability to transfer heat through a material by conduction. Note that Fourier’s law applies for all matter, regardless of its state (solid, liquid, or gas), therefore, it is also defined for liquids and gases.
The thermal conductivity of most liquids and solids varies with temperature. For vapors, it also depends upon pressure. In general:
Most materials are very nearly homogeneous, therefore we can usually write k = k (T). Similar definitions are associated with thermal conductivities in the y- and z-directions (ky, kz), but for an isotropic material the thermal conductivity is independent of the direction of transfer, kx = ky = kz = k.
Tungsten-Rhenium Alloy – Specific Heat
Specific heat of Tungsten-Rhenium Alloy is 140 J/g K.
Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The intensive properties cv and cp are defined for pure, simple compressible substances as partial derivatives of the internal energy u(T, v) and enthalpy h(T, p), respectively:
where the subscripts v and p denote the variables held fixed during differentiation. The properties cv and cp are referred to as specific heats (or heat capacities) because under certain special conditions they relate the temperature change of a system to the amount of energy added by heat transfer. Their SI units are J/kg K or J/mol K.
Properties and prices of other materials
material-table-in-8k-resolution