This article contains comparison of key thermal and atomic properties of aluminium and titanium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Aluminium vs Titanium.
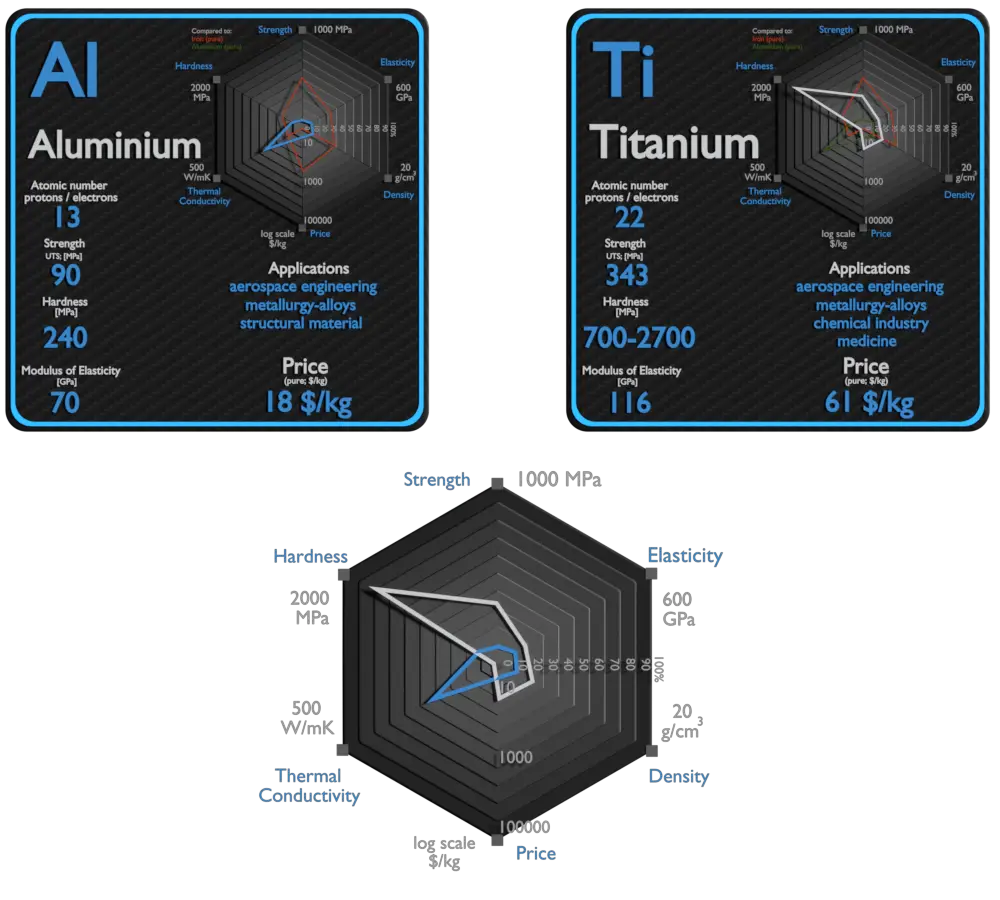
Aluminium and Titanium – About Elements


Source: www.luciteria.com
Aluminium and Titanium – Applications
Aluminium
Aluminium and its alloys are used widely in aerospace, automotive, architectural, lithographic, packaging, electrical and electronic applications. It is the prime material of construction for the aircraft industry throughout most of its history. About 70% of commercial civil aircraft airframes are made from aluminium alloys, and without aluminium civil aviation would not be economically viable. Automotive industry now includes aluminium as engine castings, wheels, radiators and increasingly as body parts. 6111 aluminium and 2008 aluminium alloy are extensively used for external automotive body panels. Cylinder blocks and crankcases are often cast made of aluminium alloys.
Titanium
The two most useful properties of the metal are corrosion resistance and strength-to-density ratio, the highest of any metallic element. The corrosion resistance of titanium alloys at normal temperatures is unusually high. These properties determine application of titanium and its alloys. The earliest production application of titanium was in 1952, for the nacelles and firewalls of the Douglas DC-7 airliner. High specific strength, good fatigue resistance and creep life, and good fracture toughness are characteristics that make titanium a preferred metal for aerospace applications. Aerospace applications, including use in both structural (airframe) components and jet engines, still account for the largest share of titanium alloy use. On the supersonic aircraft SR-71, titanium was used for 85% of the structure. Due to very high inertness, titanium has many biomedical applications, which is based on its inertness in the human body, that is, resistance to corrosion by body fluids.
Aluminium and Titanium – Comparison in Table
| Element | Aluminium | Titanium |
| Density | 2.7 g/cm3 | 4.507 g/cm3 |
| Ultimate Tensile Strength | 90 MPa (pure), 600 MPa (alloys) | 434 MPa, 293 MPa (pure) |
| Yield Strength | 11 MPa (pure), 400 MPa (alloys) | 380 MPa |
| Young’s Modulus of Elasticity | 70 GPa | 116 GPa |
| Mohs Scale | 2.8 | 6 |
| Brinell Hardness | 240 MPa | 700 – 2700 MPa |
| Vickers Hardness | 167 MPa | 800 – 3400 MPa |
| Melting Point | 660 °C | 1668 °C |
| Boiling Point | 2467 °C | 3287 °C |
| Thermal Conductivity | 237 W/mK | 21.9 W/mK |
| Thermal Expansion Coefficient | 23.1 µm/mK | 8.6 µm/mK |
| Specific Heat | 0.9 J/g K | 0.52 J/g K |
| Heat of Fusion | 10.79 kJ/mol | 15.45 kJ/mol |
| Heat of Vaporization | 293.4 kJ/mol | 421 kJ/mol |