Curium is a hard, dense, silvery metal with a relatively high melting point and boiling point for an actinide.
Protons and Neutrons in Curium
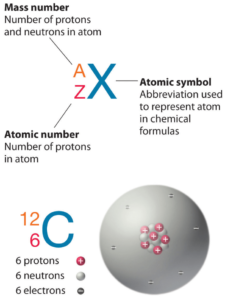 Curium is a chemical element with atomic number 96 which means there are 96 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.
Curium is a chemical element with atomic number 96 which means there are 96 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. Neutron number plus atomic number equals atomic mass number: N+Z=A. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.
For stable elements, there is usually a variety of stable isotopes. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. Mass numbers of typical isotopes of Curium are 242-250.
Main Isotopes of Curium
Curium does not occur in natural isotope.
Naturally Occuring Isotopes
| Isotope | Abundance | Neutron Number |
| – | – | – |
Typical Unstable Isotopes
| Isotope | Half-life | Decay Mode | Product |
| 242Cm | 160 d | spontaneous fission or alpha decay | – or 238Pu |
| 243Cm | 29.1 y | alpha decay or electron capture or spontaneous fission | 239Pu or 243Am or – |
| 244Cm | 18.1 y | spontaneous fission or alpha decay | – or 240Pu |
| 245Cm | 8500 y | spontaneous fission or alpha decay | – or 241Pu |
| 246Cm | 4730 y | alpha decay or spontaneous fission | 242Pu or – |
| 247Cm | 1.56×107 y | alpha decay | 243Pu |
| 248Cm | 3.40×105 y | alpha decay or spontaneous fission | 244Pu or – |
| 250Cm | 9000 y | spontaneous fission or alpha decay or beta decay | – or 246Pu or 250Bk |
Electrons and Electron Configuration
The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. Therefore, the number of electrons in neutral atom of Curium is 96. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom.
Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. The configuration of these electrons follows from the principles of quantum mechanics. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. In the periodic table, the elements are listed in order of increasing atomic number Z.
Electron configuration of Curium is [Rn] 5f7 6d 7s2.
Possible oxidation states are +3.
Summary
| Element | Curium |
| Number of protons | 96 |
| Number of neutrons (typical isotopes) | 242-250 |
| Number of electrons | 96 |
| Electron configuration | [Rn] 5f7 6d 7s2 |
| Oxidation states | +3 |
Source: www.luciteria.com