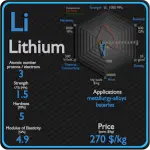
This article contains comparison of key thermal and atomic properties of chlorine and calcium, two comparable chemical elements from the periodic table. It also contains basic descriptions and applications of both elements. Chlorine vs Calcium.

Chlorine and Calcium – About Elements


Source: www.luciteria.com
Chlorine and Calcium – Applications
Chlorine
Chlorine is used in the manufacture of a wide range of consumer products, about two-thirds of them organic chemicals such as polyvinyl chloride (PVC), many intermediates for the production of plastics, and other end products which do not contain the element. As a common disinfectant, elemental chlorine and chlorine-generating compounds are used more directly in swimming pools to keep them sanitary. While perhaps best known for its role in providing clean drinking water, chlorine chemistry also helps provide energy-efficient building materials, electronics, fiber optics, solar energy cells, 93 percent of life-saving pharmaceuticals, 86 percent of crop protection compounds, medical plastics, and much more.
Calcium
The largest use of metallic calcium is in steelmaking, due to its strong chemical affinity for oxygen and sulfur. Its oxides and sulfides, once formed, give liquid lime aluminate and sulfide inclusions in steel which float out. Calcium compounds are used as manufacture of insecticides, paints, blackboard chalk, textile and fireworks.
Chlorine and Calcium – Comparison in Table
| Element | Chlorine | Calcium |
| Density | 0.0032 g/cm3 | 1.55 g/cm3 |
| Ultimate Tensile Strength | N/A | 110 MPa |
| Yield Strength | N/A | N/A |
| Young’s Modulus of Elasticity | N/A | 20 GPa |
| Mohs Scale | N/A | 1.5 |
| Brinell Hardness | N/A | 170 – 400 MPa |
| Vickers Hardness | N/A | N/A |
| Melting Point | -101 °C | 842 °C |
| Boiling Point | -34.6 °C | 1484 °C |
| Thermal Conductivity | 0.0089 W/mK | 200 W/mK |
| Thermal Expansion Coefficient | N/A | 22.3 µm/mK |
| Specific Heat | 0.48 J/g K | 0.63 J/g K |
| Heat of Fusion | 3.23 kJ/mol | 8.54 kJ/mol |
| Heat of Vaporization | 10.2 kJ/mol | 153.3 kJ/mol |