About Selenium
Selenium is a nonmetal with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, and also has similarities to arsenic. It rarely occurs in its elemental state or as pure ore compounds in the Earth’s crust.
Summary
| Element | Selenium |
| Atomic number | 34 |
| Element category | Non Metal |
| Phase at STP | Solid |
| Density | 4.819 g/cm3 |
| Ultimate Tensile Strength | 300 MPa |
| Yield Strength | 150 MPa |
| Young’s Modulus of Elasticity | 10 GPa |
| Mohs Scale | 2 |
| Brinell Hardness | 740 MPa |
| Vickers Hardness | N/A |
| Melting Point | 221 °C |
| Boiling Point | 685 °C |
| Thermal Conductivity | 2.04 W/mK |
| Thermal Expansion Coefficient | 37 µm/mK |
| Specific Heat | 0.32 J/g K |
| Heat of Fusion | 6.694 kJ/mol |
| Heat of Vaporization | 37.7 kJ/mol |
| Electrical resistivity [nanoOhm meter] | — |
| Magnetic Susceptibility | −25e-6 cm^3/mol |
Applications of Selenium
The chief commercial uses for selenium today are glassmaking and pigments. Selenium finds applications in the various industries, for example, solar cells and photoconductor applications, manganese electrolysis, DC power surge protection or X-ray crystallography.
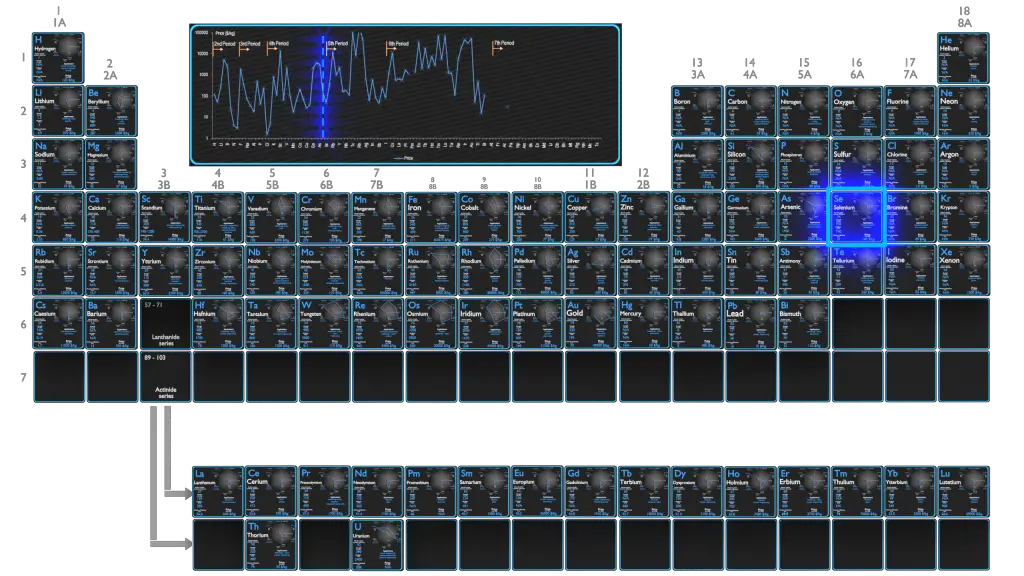
Production and Price of Selenium
Raw materials prices change daily. They are primarily driven by supply, demand and energy prices. In 2019, prices of pure Selenium were at around 140 $/kg.
Selenium is most commonly produced from selenide in many sulfide ores, such as those of copper, nickel, or lead. Electrolytic metal refining is particularly productive of selenium as a byproduct, obtained from the anode mud of copper refineries. About 2,000 tonnes of selenium were produced in 2011 worldwide, mostly in Germany (650 t), Japan (630 t), Belgium (200 t), and Russia (140 t).
Source: www.luciteria.com
Mechanical Properties of Selenium
Strength of Selenium
In mechanics of materials, the strength of a material is its ability to withstand an applied load without failure or plastic deformation. Strength of materials basically considers the relationship between the external loads applied to a material and the resulting deformation or change in material dimensions. In designing structures and machines, it is important to consider these factors, in order that the material selected will have adequate strength to resist applied loads or forces and retain its original shape. Strength of a material is its ability to withstand this applied load without failure or plastic deformation.
For tensile stress, the capacity of a material or structure to withstand loads tending to elongate is known as ultimate tensile strength (UTS). Yield strength or yield stress is the material property defined as the stress at which a material begins to deform plastically whereas yield point is the point where nonlinear (elastic + plastic) deformation begins.
See also: Strength of Materials
Ultimate Tensile Strength of Selenium
Ultimate tensile strength of Selenium is 300 MPa.
Yield Strength of Selenium
Yield strength of Selenium is 150 MPa.
Modulus of Elasticity of Selenium
The Young’s modulus of elasticity of Selenium is 10 GPa.
Hardness of Selenium
In materials science, hardness is the ability to withstand surface indentation (localized plastic deformation) and scratching. Brinell hardness test is one of indentation hardness tests, that has been developed for hardness testing. In Brinell tests, a hard, spherical indenter is forced under a specific load into the surface of the metal to be tested.
Brinell hardness of Selenium is approximately 740 MPa.
The Vickers hardness test method was developed by Robert L. Smith and George E. Sandland at Vickers Ltd as an alternative to the Brinell method to measure the hardness of materials. The Vickers hardness test method can be also used as a microhardness test method, which is mostly used for small parts, thin sections, or case depth work.
Vickers hardness of Selenium is approximately N/A.
Scratch hardness is the measure of how resistant a sample is to permanent plastic deformation due to friction from a sharp object. The most common scale for this qualitative test is Mohs scale, which is used in mineralogy. The Mohs scale of mineral hardness is based on the ability of one natural sample of mineral to scratch another mineral visibly.
Selenium is has a hardness of approximately 2.
See also: Hardness of Materials
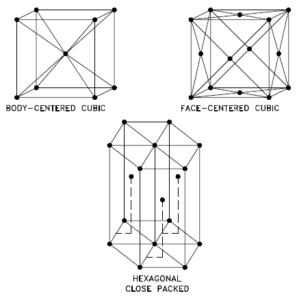
Selenium – Crystal Structure
A possible crystal structure of Selenium is hexagonal structure.
In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. The forces of chemical bonding causes this repetition. It is this repeated pattern which control properties like strength, ductility, density, conductivity (property of conducting or transmitting heat, electricity, etc.), and shape. There are 14 general types of such patterns known as Bravais lattices.
See also: Crystal Structure of Materials
Crystal Structure of Selenium

Thermal Properties of Selenium
Selenium – Melting Point and Boiling Point
Melting point of Selenium is 221°C.
Boiling point of Selenium is 685°C.
Note that, these points are associated with the standard atmospheric pressure.
Selenium – Thermal Conductivity
Thermal conductivity of Selenium is 2.04 W/(m·K).
The heat transfer characteristics of a solid material are measured by a property called the thermal conductivity, k (or λ), measured in W/m.K. It is a measure of a substance’s ability to transfer heat through a material by conduction. Note that Fourier’s law applies for all matter, regardless of its state (solid, liquid, or gas), therefore, it is also defined for liquids and gases.
Coefficient of Thermal Expansion of Selenium
Linear thermal expansion coefficient of Selenium is 37 µm/(m·K)
Thermal expansion is generally the tendency of matter to change its dimensions in response to a change in temperature. It is usually expressed as a fractional change in length or volume per unit temperature change.
Selenium – Specific Heat, Latent Heat of Fusion, Latent Heat of Vaporization
Specific heat of Selenium is 0.32 J/g K.
Heat capacity is an extensive property of matter, meaning it is proportional to the size of the system. Heat capacity C has the unit of energy per degree or energy per kelvin. When expressing the same phenomenon as an intensive property, the heat capacity is divided by the amount of substance, mass, or volume, thus the quantity is independent of the size or extent of the sample.
Latent Heat of Fusion of Selenium is 6.694 kJ/mol.
Latent Heat of Vaporization of Selenium is 37.7 kJ/mol.
Latent heat is the amount of heat added to or removed from a substance to produce a change in phase. This energy breaks down the intermolecular attractive forces, and also must provide the energy necessary to expand the gas (the pΔV work). When latent heat is added, no temperature change occurs. The enthalpy of vaporization is a function of the pressure at which that transformation takes place.
Selenium – Electrical Resistivity – Magnetic Susceptibility
Electrical property refers to the response of a material to an applied electric field. One of the principal characteristics of materials is their ability (or lack of ability) to conduct electrical current. Indeed, materials are classified by this property, that is, they are divided into conductors, semiconductors, and nonconductors.
See also: Electrical Properties
Magnetic property refers to the response of a material to an applied magnetic field. The macroscopic magnetic properties of a material are a consequence of interactions between an external magnetic field and the magnetic dipole moments of the constituent atoms. Different materials react to the application of magnetic field differently.
See also: Magnetic Properties
Electrical Resistivity of Selenium
Electrical resistivity of Selenium is — nΩ⋅m.
Electrical conductivity and its converse, electrical resistivity, is a fundamental property of a material that quantifies how Selenium conducts the flow of electric current. Electrical conductivity or specific conductance is the reciprocal of electrical resistivity.
Magnetic Susceptibility of Selenium
Magnetic susceptibility of Selenium is −25e-6 cm^3/mol.
In electromagnetism, magnetic susceptibility is the measure of the magnetization of a substance. Magnetic susceptibility is a dimensionless proportionality factor that indicates the degree of magnetization of Selenium in response to an applied magnetic field.